


Next: Design Considerations
Up: An Ontology of Materials
Previous: Ontology of materials
Almost no industry branch has developed as quickly as the polymer
industry in the last decades. Many properties of new developed
polymers showed to be ideal for the tasks of the matrix in polymer
composites. Thus, progress in composite technology was mainly linked to
improvements in polymer chemistry. The main target was the development
of new high temperature plastics-especially thermoplastics-with
outstanding chemical and mechanical properties.
Polymers can be classified under four different aspects, like:
- Manufacturing process
- polymerisation
- The chemical conversion of one or several
chemically different and active low-molecular connections (monomers)
to a linear branched, meshed or cyclic polymer. The most common
polymerisation is the radical polymerisation.
- polycondensation
- Chemical reaction where bi- or higher
functional low-molecular connections (monomers) are converted to
linear or meshed macromolecular substances (polycondensates) by
proceeeding condensation reactions. This is always combined with a
separation (condensation) of low-molecular products, like water,
ammonia, hydrochlorgas and methanol.
- copolymerisation (polyaddition)
- With copolymerisation, monomers are
directly added to the basic structure, in contrast to a
polycondensation, where low-molecular parts are separated.
- Origin of raw materials
- modified natural materials
- Materials which originate from a
natural material and have been modified for an industrial use.
- synthetic materials
- Materials which do not originate from a
natural material and have been produced artficially.
- Composition of the macromolecules
- amorphous structure
- Macromolecular design of a material, which
only shows a short range order of the atoms (e.g. glass) in contrast
to a wide range order of cristalline structures.
- semi-cristalline structure
- Materials with a partial wide range
order of atoms but also a coexisting irregular, amorphous structure.
- liquid-cristalline structure
- An intermediate state between the
amorphous and cristalline state of high-molecular polymers, which
are liquid but nevertheless have a regular, cristalline molecular
structure.
- Processing technology and polymer properties
- thermoplastics
- Polymers with a loose connection of the molecule
chains to each other. They can be melt open again by heat.
- thermosets
- Polymers with a fixed meshed net of polymer chains
to one big molecule which cannot be separated again after the
meshing reaction (curing).
- elastomers
- Polymers with irregular orientated molecule chains,
which in contrast to thermoplastics and thermosets are able to move
slightly against each other. After a deformation they automatically return to
their initial position.
In contrast to the general subdivision of materials, the above main
criteria are not unequivocal, i.e. a polymer is not belonging to only
one single group but needs to be classified by all of them.
The resulting ontology is sketched in figure 7.
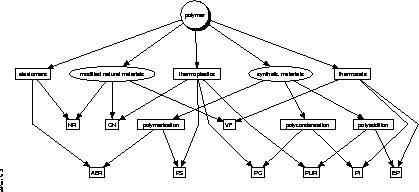
Figure 7: The plastics ontology
The (9) leaves of the hierarchy do by no way represent the complete
set of existing polymers with fixed properties and chemical
composition .
However, each of them may be regarded as a representative of the
possible combination of the characteristics 1, 2, and
4 as mentioned above. Hence, we omitted characteristic 3
since it would complicate the layout of the hierarchy and it is not
important for our application. The pure definition of the classes
without properties can be found in appendix C.
.
However, each of them may be regarded as a representative of the
possible combination of the characteristics 1, 2, and
4 as mentioned above. Hence, we omitted characteristic 3
since it would complicate the layout of the hierarchy and it is not
important for our application. The pure definition of the classes
without properties can be found in appendix C.



Next: Design Considerations
Up: An Ontology of Materials
Previous: Ontology of materials
Anna-Maria Schoeller
Mon Nov 20 15:18:31 MET 1995


![]() .
However, each of them may be regarded as a representative of the
possible combination of the characteristics 1, 2, and
4 as mentioned above. Hence, we omitted characteristic 3
since it would complicate the layout of the hierarchy and it is not
important for our application. The pure definition of the classes
without properties can be found in appendix C.
.
However, each of them may be regarded as a representative of the
possible combination of the characteristics 1, 2, and
4 as mentioned above. Hence, we omitted characteristic 3
since it would complicate the layout of the hierarchy and it is not
important for our application. The pure definition of the classes
without properties can be found in appendix C.